| Biological Information | |
|---|---|
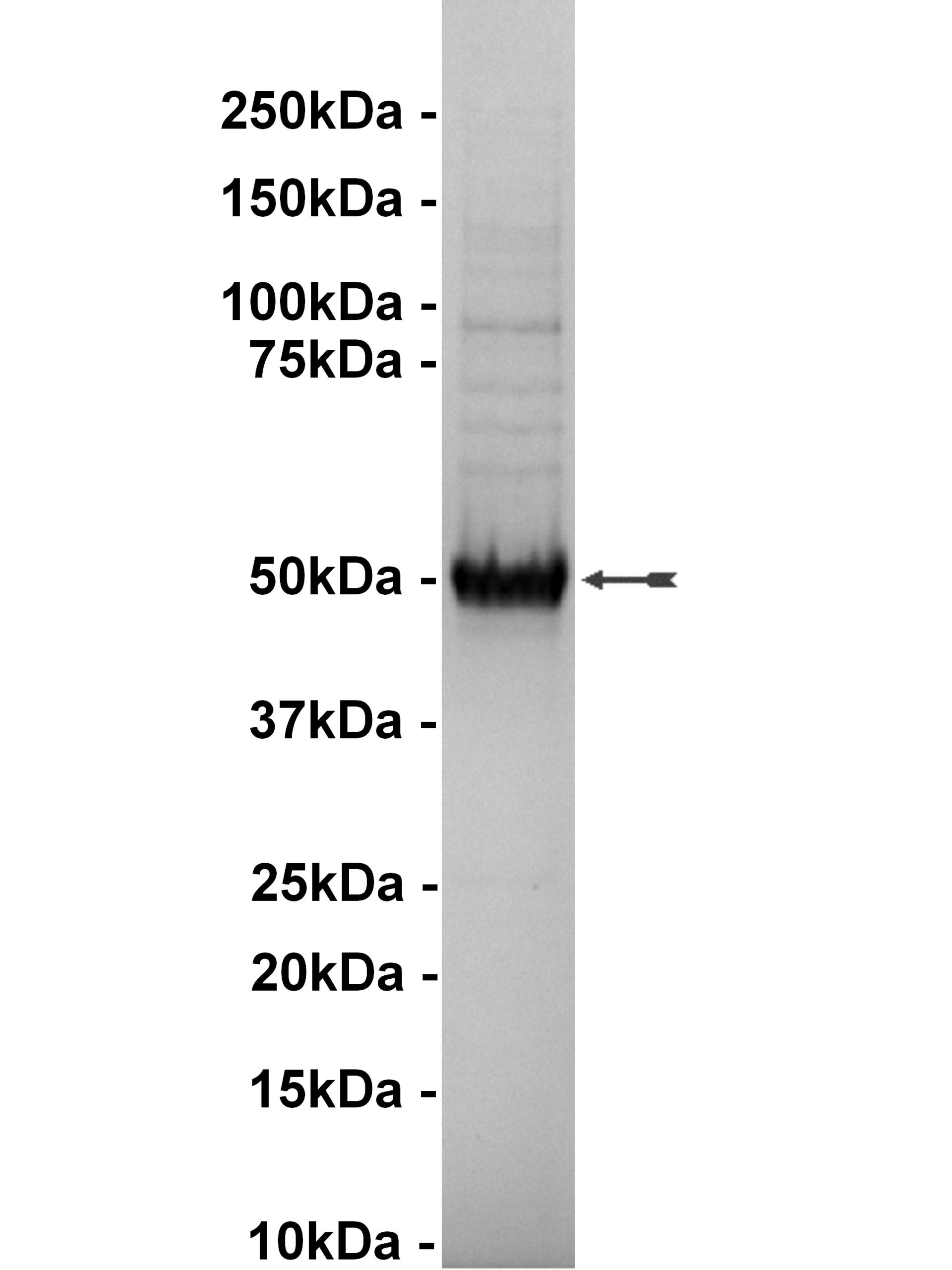
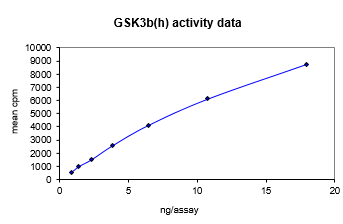
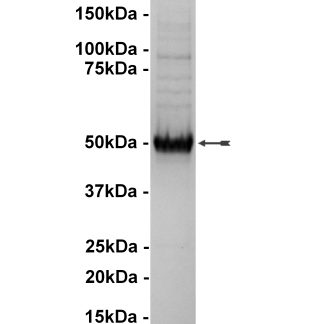
| Background Information: | Glycogen synthase kinase-3 (GSK-3) is a protein kinase that was originally identified as a regulator of glycogen synthase, a key enzyme in glycogen metabolism. There are two isoforms of GSK-3, GSK-3a and GSK-3b, and they are strictly regulated via phosphorylation. Phosphorylation of GSK-3b on Ser9 by protein kinase B (PKB) causes its inactivation and is the primary mechanism responsible for growth factor inhibition of this kinase. Activation of GSK-3b is dependent upon the phosphorylation of Tyr216. Upon activation, it has been shown to phosphorylate a number of different cellular proteins, including p53, c-Myc, c-Jun, heat shock factor-1 (HSF-1), and cyclin D1. GSK-3b also has been shown to phosphorylate aberrant sites on the microtubule associated protein tau, which is critical for the progression of Alzheimer's disease. The inhibitors may have therapeutic opportunities for the treatment of diabetes, Alzheimer's disease, stroke and bipolar disorder.
EMBL L33801, possessing the amino acid residue substitution H350L. This conflict is reported in the GenBank EST’s AI761503 and AL521664, and the March updated version of Swiss Prot P49841. |
| Target Class: | Kinase |
| Accession Number: | NM_002093.2 |
| Target Name: | GSK3β |
| Target Aliases: | GSK3B |
| Target Species: | Human |
| Usage | |
| Product Type: | Enzymes |
| Application: | Drug Discovery & Development |
| Storage Conditions: | 6 months at -70°C |
| Usage Disclaimer: | These products may be covered by issued US and/or foreign patents, patent application and subject to Limited Use Label License. Please visit discoverx.com/license for a list of products that are governed by limited use label license terms and relevant patent and trademark information. |
Datasheets
14-306x Datasheet
View Document